

 |  |
PERFORMING CAROTID STENTING FOR STROKE PREVENTION: Minimally Invasive Procedure Is Promising New Option
Stroke is the most common cause of death and the number one cause of disability in adults in the United States. The American Heart Association estimates that 20% to 30% of strokes are caused by particles of atherosclerotic plaque, which travel upstream into the vessels — the pair of carotid arteries in the neck — that supply the brain. Approximately 730,000 strokes are reported each year in the United States, and approximately 150,000 of these events result in death.
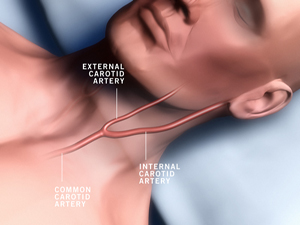
![]() Many of these life-threatening strokes can be prevented. For patients with blocked carotid arteries, carotid endarterectomy (removal of plaque) has long been the standard surgical treatment. However, although carotid endarterectomy is well established, the need for therapeutic options is important for certain patients who are not good surgical candidates for it.
Many of these life-threatening strokes can be prevented. For patients with blocked carotid arteries, carotid endarterectomy (removal of plaque) has long been the standard surgical treatment. However, although carotid endarterectomy is well established, the need for therapeutic options is important for certain patients who are not good surgical candidates for it.
At Stony Brook, in addition to carotid endarterectomy, our vascular surgeons perform carotid stenting that offers a promising new treatment option. The "endovascular" (within the vessel) operation is minimally invasive, and in selected patients it has the potential to be performed as a same-day procedure.
Our multidisciplinary team includes vascular surgeons, cardiologists, neurologists, and neuroradiologists who evaluate each patient and discuss each potential case for stenting. This team approach helps to ensure and distinguish the quality of care that patients receive at the University Vascular Center at Stony Brook.
As we age, our arteries narrow. They lose their flexibility and the linings thicken. This process is called atherosclerosis or "hardening of the arteries." When the carotid arteries in the neck begin to narrow, blood clots may develop. These arteries may become completely blocked or a piece of clot may break off and travel to the brain. In both cases a stroke — also known as "brain attack" — may result. Transient ischemic attacks ("mini-strokes") may happen first.
Risk of stroke increases as the carotid arteries become more and more narrow. The risk is particularly high after the arteries are blocked more than 70%. The amount of blockage may be determined by a simple painless test called a carotid doppler or carotid ultrasound. People at greatest risk for carotid artery narrowing are those over age 65 (particularly smokers) and those who already have poor circulation in the legs or their heart. Patients who have temporary loss of vision or speech and/or weakening of an arm or leg may have had a mini-stroke and should see their doctor.
CAROTID STENTING
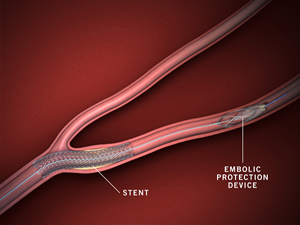
![]() Carotid stenting is an endovascular procedure in which a tiny, slender metal-mesh tube (stent) is fitted inside a carotid artery to increase the flow of blood blocked by plaques. The stent is inserted in the carotid artery across the blockage and a balloon is threaded up to the blocked artery, and then inflated so that it presses against the plaque, flattening it and re-opening the artery.
Carotid stenting is an endovascular procedure in which a tiny, slender metal-mesh tube (stent) is fitted inside a carotid artery to increase the flow of blood blocked by plaques. The stent is inserted in the carotid artery across the blockage and a balloon is threaded up to the blocked artery, and then inflated so that it presses against the plaque, flattening it and re-opening the artery.
This is all done through a puncture in the groin. The stent acts like a scaffold to prevent the artery from collapsing or being closed by plaque after the procedure is completed.
Stenting is recommended for patients who are unable to undergo carotid endarterectomy, which is the standard treatment for severe buildup of plaque in the carotid artery, and which is the most common vascular surgery performed in the United States today. But for some patients, it may not be appropriate, such as those who cannot tolerate the side effects of anesthesia.
Carotid stenting, though generally safe and less invasive than endarterectomy, offers a promising alternative. It is currently being refined as a procedure, in terms of both technique and the stents themselves.
The first randomized controlled trial comparing surgery to carotid angioplasty and stenting was reported at the American Heart Association in 2002, and showed significant benefits of stenting over surgery in reducing adverse events at one month in "high risk" patients. Subsequent studies have confirmed its effectiveness and safety.
FDA APPROVAL
In 2004, the FDA approved the Guidant stent and protection device for use in treating carotid artery blockages. The device is approved for use in patients who are at "high risk" for carotid endarterectomy, and who have had lateralizing cerebrovascular symptoms and a stenosis of at least 50% by angiography or duplex ultrasound.
The FDA also approved this device for asymptomatic patients whose carotid artery is at least 80% stenotic by angiogram or duplex ultrasound, and who are also "high-risk" candidates for carotid endarterectomy.